Effect of USP Sample Storage Conditions on Microbial Recovery from Tryptic Soy Agar (TSA) Plates
Deanna L. Kiska1, Rebecca A. Moran1, and Chris Lee2
1U.S. Micro-Solutions, Inc., Latrobe, PA and 2EpiClear Consulting, Columbus, OH
Introduction
The United States Pharmacopeia (USP) – National Formulary (NF) has established minimum standards for the control and monitoring of sterile compounding facilities and aseptic processing environments, e.g., USP <797>, USP <1116>. These standards require viable air and surface sampling using an appropriate agar medium, e.g., TSA, which allows growth of bacterial and fungal organisms. Recommended action levels, based on the number of colony forming units (CFU), have been established for each sample type and ISO class. Conditions that negatively affect microorganism viability could seriously impact interpretation of these cultures.
Shipping sampled agar plates to an outside laboratory for analysis is a common practice. While the recommended practice is to ship samples on the day of collection, this is not possible for samples that are collected late in the day or on Fridays. Some laboratories are not open on Saturday so samples collected on Friday are stored until Monday for shipping. Since there is a dearth of information on the effect of storage conditions on microbial recovery from USP samples, our study sought to evaluate the impact of room temperature and refrigerated storage on the viability of several bacterial and fungal organisms on TSA media.
Methods
Standardized suspensions of 6 bacterial and fungal organisms were inoculated to TSA plates. The organisms included Aspergillus brasiliensis (AB), Candida albicans (CA), Bacillus subtilis (BS), Escherichia coli (EC), Staphylococcus aureus (SA), and Pseudomonas aeruginosa (PA).
Plates were held at 2-8oC (Refrigerated) and at 20-21oC (Room temperature). After 24, 48, and 72 hours at each storage temperature, the plates were incubated at room temperature for 24 hours to simulate overnight shipping and then were placed at 30-35oC for 48-72 hours with colony growth counted daily. The results were compared to control plates that were incubated immediately after inoculation.
Statistical analysis involved three multivariate regression models that were created to explore the impact of refrigeration and room temperature exposure of samples prior to the shipping process on the final microbial counts of the samples after incubation.
Results
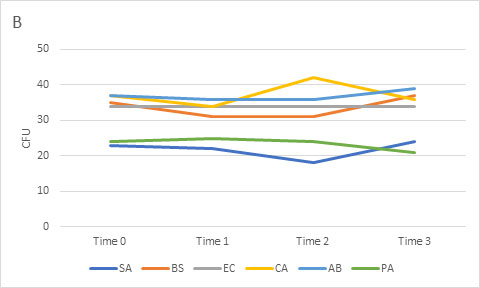
Overall, samples that were refrigerated yielded significantly lower microbial counts than samples that were exposed to room temperature conditions, with each subsequent day of refrigeration significantly decreasing the final count. When looking at each organism individually, there was minimal effect of refrigeration on B. subtilis and E. coli but substantial effects on the remaining bacterial and fungal organisms (Figure).
Samples that were exposed to room temperature storage conditions did not yield microbial counts that were significantly different from the control samples at any time point. The length of room temperature exposure (up to 3 days) did not significantly impact the counts (Figure). During room temperature storage, organism growth was noted at 24 hours for E. coli, with the remaining organisms showing growth by 48 hours, except for A. brasiliensis at 72 hours. Although no growth of A. brasiliensis was noted on plates stored for 48 hours, growth subsequently occurred during the 24-hour simulated shipping time. Sporulation during storage or shipping could cause fungal spores to be dispersed, resulting in falsely high counts and possible contamination of other plates if the plates are not rim sealed.
Based on these results, we recommend not more than 24 hours of room temperature storage prior to shipping. The plates should be rim sealed, placed in sealed sterile plastic bags, and kept at room temperature (~21oC/~70oF) out of direct sunlight and away from HVAC vents. The temperature should remain fairly consistent throughout the storage time to prevent fluctuations which could cause plate condensation. As per our previous shipping study, plates should be shipped at ambient temperature, preferably in a Styrofoam-lined box which prevents exposure to wide temperature fluctuations and provides additional cushioning to prevent plate breakage.
U.S. Micro Solutions, Inc.
U.S. Micro Solutions, Inc. U.S. Micro-Solutions, Inc. is a full-service, ISO 17025 accredited, CGMP-compliant environmental microbiology laboratory located in Latrobe, PA. Whether you are a cleanroom certifier, pharmacist, water treatment professional, industrial hygienist, or mold remediation specialist, we can assist you with choosing the right sample and the right test to meet your specific needs.
You can trust U.S. Micro-Solutions to deliver quality results, fast turn-around time, and exceptional customer service. Call us today at 724-853-4047, or visit our Contact Us page – we are here to help!
Request a Copy of
the Full Study
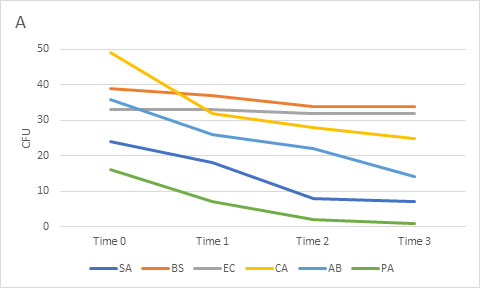
Mean colony count viability (CFU) for refrigerated (A) and room temperature (B) samples